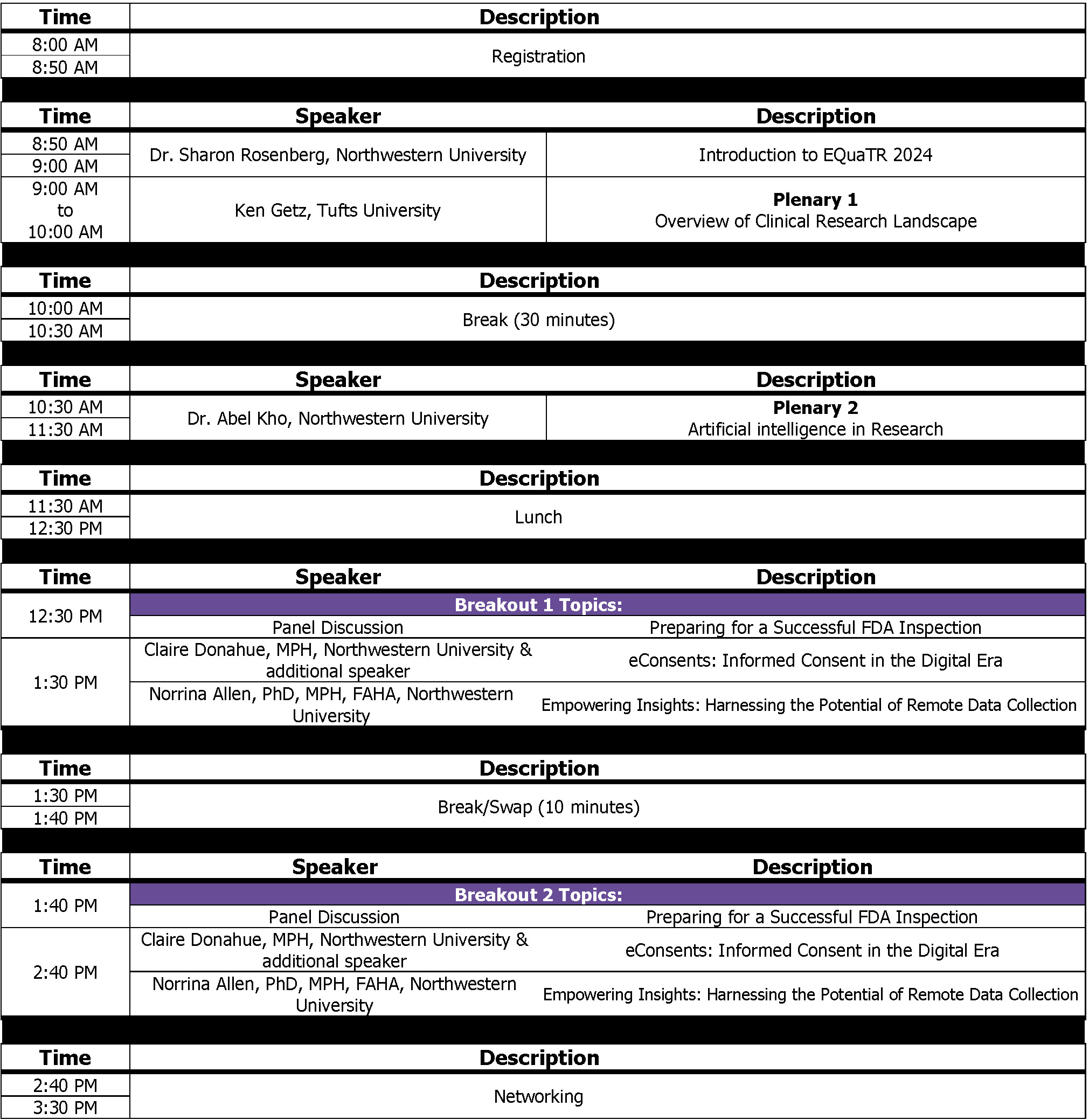
Plenary Speakers
Overview of Clinical Research Landscape
Ken Getz
Executive Director and Professor | Tufts Center for the Study of Drug Development
Tufts University School of Medicine
Founder and Board Chair of CISCRP
Artificial intelligence in Research
Dr. Abel Kho
Director, Institute for Public Health and Medicine (IPHAM) - Center for Health Information Partnerships
Director, Institute for Artificial Intelligence in Medicine
Professor of Medicine (General Internal Medicine) and Preventive Medicine (Health and Biomedical Informatics)
Session Speakers
Panel discussion: Preparing for a Successful FDA Inspection
Moderator
Briana Harrison
Clinical Research Manager, University of Chicago
Panelists
Sara Moellering
Director, Clinical Research Operations, University of Chicago
Susan Muhr Leister
Vice President of Quality and Compliance, Technical Resources International
Jeanne Thai, MPA
Investigator, OBIMO/Division of Bioresearch Monitoring Operations WEST, Office of Regulatory Affairs, Chicago District Office, U.S. Food and Drug Administration
eConsents: Informed Consent in the Digital Era
Claire Donahue, MPH
Clinical Operations Manager, Clinical Trials Office
Robert H. Lurie Comprehensive Cancer Center
Northwestern University
and
Aditya Sanzgiri
Director – Research Compliance
Office of Clinical Research
Biological Sciences Division
University of Chicago
Empowering Insights: Harnessing the Potential of Remote Data Collection
Dr. Norrina Allen, PhD, MPH, FAHA
Quentin D. Young Professor of Health Policy
Professor of Epidemiology and Pediatrics, Tenured
Department of Preventive Medicine
Feinberg School of Medicine
Northwestern University
Director, Center for Epidemiology and Population Health
Institute for Public Health and Medicine (IPHAM)
Co-Director of the Data Science Hub
Institute for Innovations in Developmental Sciences (DevSci)